Background
In 2018, the Governance Committee for the implementation of the Government-wide Scientific Integrity PolicyFootnote 1 (the Governance Committee), whose membership includes the Chief Science Advisor, the Secretary of the Treasury Board (TB), and the President of the Professional Institute of the Public Service of Canada (PIPSC), published the Model Policy on Scientific Integrity (the model policy)Footnote 2. The model was drafted as a guide for 24 (now 25Footnote 3) federal departments and agencies that were required to implement a scientific integrity policy (SIP) under the terms of two collective agreements negotiated between PIPSC and TB in May and June of 2017. Since January 2020, implicated departments and agencies have been surveyed annually to determine their progress in implementing their scientific integrity policies and procedures. Results of the 2020 and 2021 surveys were reported in The Status of Federal Scientific Integrity Policies – February 2021Footnote 4 report, published in 2021. Results of the 2022Footnote 5 and 2023Footnote 6 surveys have been published in subsequent reports. The current report summarizes results from the fifth annual survey conducted between December 2023 and late February 2024.
The 2024 survey
The 2024 survey requested information on 15 compliance measures, each of which is associated with a non-discretionary provision of the model policyFootnote 7. Since departments and agencies may adopt, adapt or even replace the model policy as they see fit, some compliance measures may not apply if, for example, a department or agency chose to eliminate the corresponding non-discretionary provision or made it discretionary in their policy.
The 2020 and 2021 surveys included thirteen compliance measures. For the 2022 survey, a fourteenth measure providing information on progress made in developing SIP performance monitoring and evaluation plans was added. In the 2023 survey, a fifteenth compliance measure providing information on measures in place to support training on evidence-informed decision-making was added.
Like the 2023 survey, the 2024 survey also included subsidiary questions associated with three original compliance measures. These subsidiary questions were designed to provide more information on employee notification about the departmental scientific integrity policy, Research Ethics Boards (REBs) and breach investigation procedures. The 2024 survey also included questions about potential amendments to the departmental policy and implementation actions taken by departments and agencies during the previous year.
As in the past, the 2024 report is based on a thorough review of all evidence provided by departments and agencies to support survey responses. Where the evidence provided was deemed insufficient to justify the submitted response, departments and agencies were contacted to discuss their responses and evidence. In some instances, these discussions resulted in revised survey responses.
2024 results
All 25 departments or agencies that are required to implement a scientific integrity policy responded to the survey. Twenty-four departments and agencies (representing 2,722 RE employees and 10,386 SP employeesFootnote 8, respectively) now have approved policies, 23 (representing 2,720 RE employees and 10,351 SP employees, respectively) of which are in effect (Fig. 1). One department reported that their policy is still in development.
Figure 1
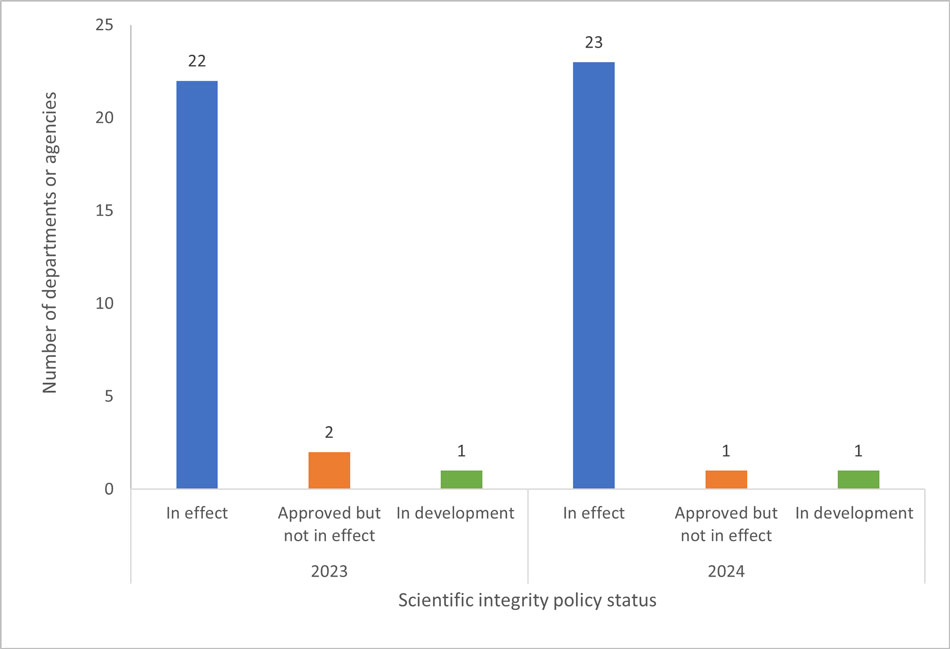
Fig. 1. The number of federal departments or agencies with scientific integrity policies in effect, approved but not in effect, or in development (i.e., not yet submitted for approval) as of January 2023 and January 2024.
-
Figure 1 - Text version
The bar graph shows the number of federal departments or agencies (DDAAs) with scientific integrity policies in effect, approved but still not in effect, or in development in 2023 and in 2024. For both years, the highest number is scientific integrity policies in effect, which rose from 22 in 2023 to 23 in 2024. DDAAs whose scientific integrity policies were approved but not in effect decreased from two in 2023 to one in 2024. One department reported their scientific integrity policy is in development in 2023 and 2024.
The model policy includes two key provisions in support of responsible conduct of research: peer review of all technical communications and, where required, approval of proposed research by a Research Ethics BoardFootnote 9. Nineteen of the 24 departments and agencies with approved SIPs have peer review requirements in placeFootnote 10. Three departments and agencies reported that implementation of peer review requirements is still pending. One department or agency elected not to report on the peer review compliance measure, and one departmentFootnote 11 considered the measure inapplicable.
Twelve departments and agencies have a process in place for ensuring that research proposals involving human subjects is reviewed by an REB, while three departments and agencies reported that implementation of an REB approval process is still pending. Two departments elected not to report on the REB compliance measure, and seven departments or agenciesFootnote 12 considered the measure to be inapplicable.
The model policy also includes non-discretionary provisions facilitating the federal science and research community in providing advice on regulations and policy, on departmental research programs, and on prioritizing investments in research.
- Seventeen of the 24 departments and agencies have processes in place for soliciting advice on policies and regulations, while five departments and agencies reported that implementation of these processes is still pending. For two departments this compliance measure is inapplicable as the corresponding provision was removed from their policy.
- Fifteen of the 24 have processes in place for soliciting advice on research programs and the prioritization of departmental research investment. Five departments and four departments reported that the implementation of processes for soliciting advice on research programs and the prioritization of departmental research investment, respectively, is still pending. For four departments, the implementation of processes for soliciting advice on research programs is inapplicable as the corresponding provision was removed from their policy. Similarly, for five departments, the implementation of processes for soliciting advice on the prioritization of departmental research investment is inapplicable as the corresponding provision was removed from their policy.
As of January 2024, the 23 departments and agencies with their SIPs approved and in effect have notified their employees about the SIP, 12 of which reported that they notify their employees about their SIP multiple times a year. The department or agency that has its SIP approved but not in effect has yet to notify its employees about the SIP.
Twenty-three departments and agencies reported they have a Scientific Integrity Lead (SIL) who is responsible for overseeing allegations of scientific integrity breach. One department, who in previous years reported having appointed a SIL, has reported in 2024 that they are yet to appoint a replacement for their previous SIL. For at least two departments and agencies, the SIL is also responsible for promoting departmental scientific integrity.
Eighteen departments and agencies have implemented a process for bringing forward allegations of scientific integrity breach. For 15 departments and agencies, this process is explicitly described in either a directive, policy, guidelines or guidance documents specific to alleged scientific integrity breaches or is part of a more general description that includes other types of alleged misconduct. For two departments and agencies, a process for bringing forward allegations of scientific integrity breach is described in a document outlining breach processes and operating procedures. One department reported that a breach process exists but is not explicitly described in a formal policy. Six departments and agencies have yet to implement a process for bringing forward allegations of scientific integrity breach.
In collaboration with the Canada School of Public Service, the OCSA has developed a set of four on-line training modules on evidence-informed decision-making to support departments and agencies in providing SIP-related training to employees. The firstFootnote 13, secondFootnote 14, thirdFootnote 15, and fourthFootnote 16 modules were launched in September 2023, November 2023, January 2024 and May 2024, respectively. Some departments and agencies began leveraging these courses, advertising them internally, and incorporating them into their employees learning plans to ensure compliance with the requirement to put in place measures to support education, training and/or professional development devoted to the roles of science and research in developing evidence to support evidence-informed decision-making. As such, 14 departments and agencies have reported compliance with this requirement in the 2024 survey (up by 9, compared to 2023 results). The remaining 10 departments and agencies reported that either they had yet to advertise the courses internally, or required more time to promote the courses and incorporate them into existing learning plans.
The 2024 survey indicates that in the six years since the model policy was developed, departments and agencies have generally made good progress on implementation of their departmental SIPs (Fig 2). However, some significant gaps remain.
- Six departments and agencies still do not have a process in effect for bringing forward and investigating breaches of scientific integrity.
- Seven departments and agencies have yet to implement measures to support education, training and/or professional development in responsible conduct in research; research ethics; and the annotation, management and archiving of research and scientific data.
- Seven departments and agencies have yet to implement processes for notifying external collaborators and contractors about SIPs.
- Most significantly, nineteen departments or agencies have yet to implement a monitoring plan that provides information on the extent to which their policy has achieved its objectives (Fig 2).
Figure 2
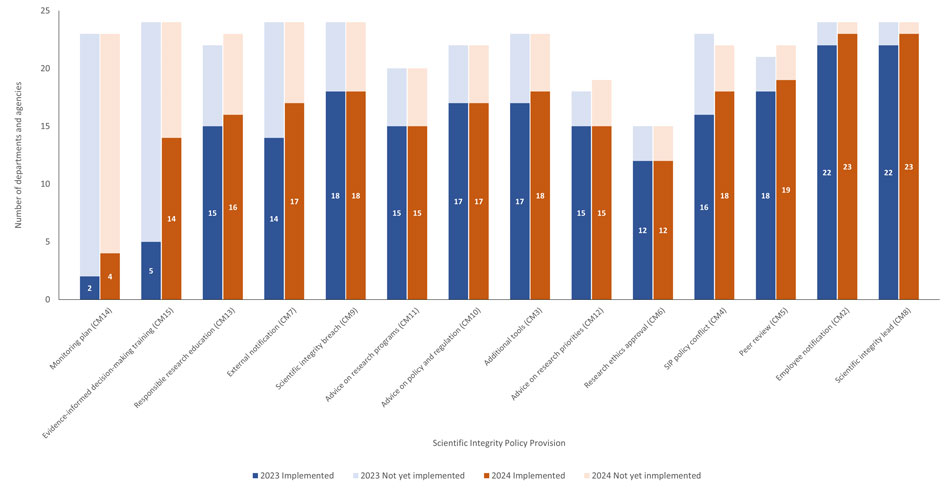
Fig. 2. The number of departments or agencies with an approved scientific integrity policy (SIP) who have or have not yet implemented the actions or activities corresponding to a particular non-discretionary provision in the model policy as of January 30, 2023 and January 30, 2024. For each compliance measure, departments and agencies with approved SIPs but (a) whose policy does not include the provision in question; or (b) who decided not to report on the associated compliance measure in the 2023Footnote 17 and 2024Footnote 18 surveys, have been excluded from the analysis. For a description of the listed compliance measures, see Appendix B.
-
Figure 2 - Text version
The stacked bar graph shows the number of departments and agencies (DDAAs) with approved scientific integrity policies that have implemented or are yet to implement various scientific integrity policy provisions, and how the number of DDAAs changed from 2023 to 2024. The provision on SIP monitoring plan is the lowest at 2 DDAAs in 2023 and 4 DDAAs in 2024, followed by evidence-informed decision-making training with the second lowest at 5 DDAAs in 2023 and 14 DDAAs in 2024. Provisions on employee notification and on appointing a scientific integrity lead have the highest number of DDAAs, both at 22 DDAAs in 2023, and both at 23 DDAAs in 2024. The largest growth in implementation was in evidence-informed decision-making training, with the number of DDAAs who implemented the provision increasing from 5 in 2023 to 14 in 2024. None of the proportions shrank from 2023 to 2024.
During the past year, two departments/agencies have amended their SIPs to include departmental specific references, position titles and contexts; referencing relevant legislations and policies; adding more specific language around REB approvals and the communications that require peer-review. Twelve departments and agencies reported that they have produced additional procedures, policies, guidelines, tools, training, or professional development opportunities relevant to their policies. These include:
- Launching a framework on science advice governance to strengthen the role and impact of departmental science in decision-making, program delivery, and policy development, and to facilitate the implementation of a Science Strategy;
- Conducting social media campaigns to raise awareness of the SIP;
- Launching a science council to provide scientific and strategic advice to senior management from the scientific workforce on research priorities, strategies, emerging needs, gaps and partnership opportunities; and to provide a forum for increased scientific awareness and collaborative opportunities across the department/agency;
- Taking steps to re-enforce transparent, impartial, and evidence-based peer review processes;
- Launching SIP online training courses, which could be included in employee learning plan;
- Delivering internal SIP-related learning sessions, including on responsible conduct in research and how SIP applies to the department’s work;
- Developing and implementing policy or guidelines for a range of actions, including investigation of alleged breaches; external communications; and dissemination of research and scientific findings;
- Investigating scientific integrity culture and the progress of SIP implementation through the administering of employee surveys to inform and enhance future SIP implementation efforts; and
- Producing factsheets to notify/inform contractors or extramural collaborators of the SIP and to outline their responsibilities as per the department’s SIP.
Over the past year, the Office of the Chief Science Advisor has continued to support federal departments and agencies in advancing federal scientific integrity. These initiatives included:
- In consultation with departments and agencies, finalizing and publishing a SIP guidance document that assists departments and agencies in selecting members for committees investigating alleged breaches of scientific integrity;
- Providing ongoing feedback to departments and agencies on their proposed SIP performance monitoring and evaluation strategies;
- Via collaboration with the Canada School for Public Service, launching four training modules on evidence-informed decision-making, pursuant to the model policy s. 7.8; and
- Commencing preliminary consultations on the draft of Version 2.0 of the Model Policy on Scientific Integrity, which includes new provisions concerning Indigenous Knowledge, the use of generative artificial intelligence tools, research security and open science, as well as amendments that attempt to resolve several operational issues that have arisen over the last five years with regard to SIP implementation in various departments and agencies.
Next steps
The 2023-2024 survey results demonstrate that considerable progress on scientific integrity has been made by federal departments and agencies, and that most compliance measures have been met across departments and agencies. While continuing to support and monitor the progress of SIP implementation, over the past year the Governance Committee also began shifting its focus towards the evolutionFootnote 19 of the SIP. As such, the Governance Committee’s priorities for 2024-2025 are as follows:
- Complete consultations and finalize Version 2.0 of the Model Policy on Scientific Integrity; and
- Continue to monitor and provide ongoing advice on SIP-implementation, notably on the development and implementation of departmental policy performance monitoring and evaluation plans.
The Governance Committee is pleased with the progress made by federal departments and agencies on scientific integrity, and recognizes the commitment by federal departments and agencies, Ministers, and employees to ensure that government decisions are informed by scientific evidence and that Canadians are informed about important scientific issues that affect them. As work progresses on updating the model policy, the committee looks forward to continued collaboration to ensure that federal scientific integrity policies are fit to address new and evolving realities.
Appendix A: List of departments and agencies that are required to implement a scientific integrity policy
- 1. Agriculture and Agri-Food Canada
- 2. Canada Border Services Agency
- 3. Canadian Food Inspection Agency
- 4. Canadian Grain Commission
- 5. Canadian Heritage
- 6. Canadian Space Agency
- 7. Correctional Service of Canada
- 8. Crown-Indigenous Relations Northern Affairs Canada
- 9. Department of National Defence
- 10. Environment and Climate Change Canada
- 11. Fisheries and Oceans Canada
- 12. Global Affairs Canada
- 13. Health Canada
- 14. Impact Assessment Agency of Canada
- 15. Indigenous Services Canada
- 16. Infrastructure Canada
- 17. Innovation, Science and Economic Development Canada
- 18. Library and Archives Canada
- 19. National Research Council
- 20. Natural Resources Canada
- 21. Public Health Agency of Canada
- 22. Public Services and Procurement Canada
- 23. Royal Canadian Mounted Police
- 24. Statistics Canada
- 25. Transport Canada
Appendix B: The scientific integrity policy (SIP) compliance survey questions
2022 SIP Compliance Survey Follow-Up Information:
- 1. Did your department/agency complete the 2022 compliance survey?
- 2. Have there been any amendments to your departmental scientific integrity policy (SIP) since the 2022 compliance survey?
- 3. Has your department developed any additional procedures, policies, guidelines, tools, training or professional development opportunities relevant to the SIP since the 2022 survey?
Compliance Measures:
- CM1. In what year did your departmental SIP come into effect?
- CM2.
- (a) Has the department or agency notified employees about the SIP? (Corresponding mSIP article: s.7.1.1.)
- (b) Does the department or agency notify employees about the SIP multiple times per year?
- CM3. Has the department or agency implemented additional procedures, policies, guidelines, tools, training or professional development opportunities in support of the SIP? (Corresponding mSIP article: s.7.1.2.).
- CM4. Does the department or agency have a process for reporting and recording instances of policy conflict or incompatibility with the SIP? (Corresponding mSIP article: s.3.5.)
- CM5. Does the department or agency require that all technical communications undergo peer-review? (Corresponding mSIP article: s.6.1., 7.5.6., and 7.8.1.(i))
- CM6.
- (a) Does the department or agency require that, where appropriate, research or scientific projects involving humans be reviewed and approved by a Research Ethics Board (REB)? (Corresponding mSIP article: s.6.1.,7.5.6., and 7.8.1.(i), 7.8. (x)). See the Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans – TCPS 2 (2018).
- (b) Please identify the federal Research Ethic Boards (REBs) that your DDAA uses for project review and approval.
- CM7. Has the department or agency taken steps to notify/inform contractors or extramural collaborators of the DDAA SIP and encourage them to comply with its articles? (Corresponding mSIP article: s.7.1.3.)
- CM8. Has the department or agency appointed a Science Integrity Lead? (Corresponding mSIP article: s.7.2.2.2.)
- CM9.
- (a) Does the department or agency have a process in effect for bringing forward and investigating breaches of scientific integrity? (Corresponding mSIP article: s.7.2.2.3.)
- (b) This process is explicitly described:
- a policy that applies specifically to alleged breaches of scientific integrity.
- a policy that applies to alleged breaches of scientific. Integrity as well as other types of misconduct.
- Other (please specify)
- Not applicable – this process is not explicitly described in a policy.
- CM10. Does the department or agency have a mechanism and/or procedure in effect for soliciting researcher/scientist advice on departmental policies and/or regulations? (Corresponding mSIP article s.7.7.1.)
- CM11. Does the department or agency have a mechanism and/or procedure in effect for soliciting researcher/ scientist advice on departmental research programs? (Corresponding mSIP article s.7.7.2.)
- CM12. Does the department or agency have a mechanism and/or procedure in effect for systematically soliciting researcher/scientist assistance in identifying and prioritizing federal investment in research? (Corresponding mSIP article: s.7.7.3.)
- CM13. Does the department or agency have measures in effect to support education, training and/or professional development in any of the following areas: responsible conduct in research; research ethics; and the annotation, management and archiving of research and scientific data? (Corresponding mSIP article: s.7.2.1.3.)
- CM14. Does your department or agency have a monitoring plan in effect for the DDAA SIP that will provide information on the extent to which the policy has achieved its objectives (policy performance)?(Corresponding mSIP article: s.7.9.)
- CM15: Does your department or agency have measures in place to support education, training and/or professional development devoted to the roles of science and research in developing evidence to support evidence-informed decision-making. (Corresponding mSIP article: s.7.7.4.)
